
About Platelet-Rich Plasma

What is PRP?
Platelet Rich Plasma (PRP) is concentrated from blood which contains healing factors, such as white blood cells and bioactive proteins, called growth factors and stem cell markers. These cells are vital for tissue regeneration and repair. Platelets, once thought of being responsible for only clotting, have been scientifically proven to be a reservoir of these vital healing components. They work by a process called pseudopodial extention, where the cell stretches parts of its body out to attach to surfaces. They can switch between their normal (unactivated) and their attaching (activated) form.
FDA Registered
Disclaimer: The products described on this website are intended for preparing platelet rich plasma (PRP) for use as a cosmetic as defined by the FDA. Cosmetic use includes topical application of PRP to the human body to cleanse, beautify, promote attractiveness, and alter appearances. These products are not intended to diagnose, treat, cure, or prevent any disease or condition.
PRP contains VEGF, FGF, IGF, TGF-b, PDGF, EGF and other growth factors.
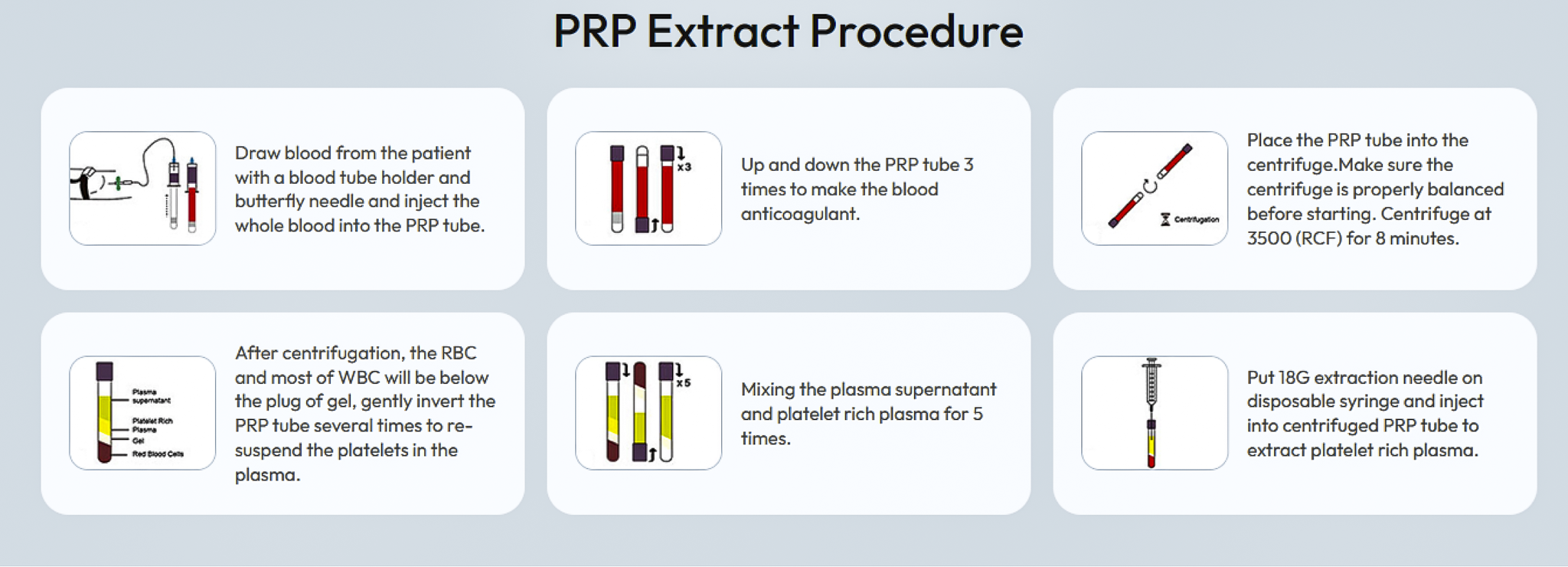
How Has PRP Technology Evolved Over Time?
Product effectiveness has been a key focus in the expanding field of PRP extraction and clinical application. PRP technology has continued to evolve as clinical demand and product design have advanced. While PRP systems vary in how they process blood and isolate platelets, many products can be grouped into three broad “generations” based on how preparation methods have developed over time:
1st Generation: The earliest generation of PRP technology was based on simple centrifugation techniques and manual preparation practices. These methods relied on standard laboratory equipment and often incorporated additives—such as activators or coagulants—to initiate platelet activation and support platelet recovery from the blood sample.
2nd Generation: Currently the most widely adopted generation in clinical settings, second-generation systems introduced purpose-built PRP kits designed to improve platelet concentration and enhance repeatability between extractions.
3rd Generation: The newest generation of PRP technology focuses on further refinement of system design and preparation protocols. These systems utilize specially developed kits engineered to maximize platelet concentration from a given blood sample while maintaining high repeatability and procedural consistency.

What Applications is PRP capable of?
PRP’s biological components initiate the body’s natural healing processes. Growth factors, immunomodulatory cells, scaffolding proteins, and extracellular vesicles influence the local microenvironment in a manner consistent with the body’s normal response to injury. This localized regenerative response may assist in wound healing, support management of chronic conditions, and potentially address tissue damage associated with degenerative disorders.
While current clinical applications of PRP remain focused on specific indications, its therapeutic potential continues to expand. As research advances and protocols become more standardized, PRP may become more widely adopted in clinical practice—offering a minimally invasive option that can support recovery in conditions that have historically been challenging to treat. In some cases, this may reduce reliance on more invasive, lengthy, or costly traditional procedures.
Research in PRP continues to grow as interest in blood-derived therapies increases. Advances in biomedical science have accelerated discoveries in regenerative and biologic treatments. Although substantial progress has been made over the past several decades, important questions remain, and ongoing investigation is necessary to further define optimal applications and outcomes.

Frequently Asked Questions
-
No. Based on current clinical evidence, PRP cannot and does not replace conventional medical or surgical treatments. While regenerative therapies continue to advance, there is no single “cure-all” approach in medicine. PRP functions by supporting the body’s natural healing processes and is therefore limited by the biological capacity for regeneration within each individual.
Established treatment methods, developed and refined over decades of clinical research, remain essential components of patient care. In some cases, PRP may complement traditional therapies or enhance treatment outcomes, but it is not a substitute for comprehensive medical management.
Although ongoing research continues to explore the expanding role of biologic therapies, the concept of a universal cure remains unlikely given the complexity of human physiology. PRP represents a promising area within regenerative medicine, yet its application is defined by the boundaries of the body’s intrinsic healing mechanisms.
-
The answer is not definitive based on current research and available evidence.
Allogeneic (same-species, different-individual) PRP applications have been explored in limited studies. Some small-scale investigations have reported regenerative responses following treatment, while also observing immune-related reactions consistent with the risks inherent to allogeneic biologic materials.
However, these studies have generally been preliminary in nature, often involving small sample sizes and, in many cases, non-human subjects. As such, their findings should be interpreted with caution. Immune response and rejection remain important considerations in any allogeneic procedure.
At present, autologous (same-individual origin) PRP remains the standard and is widely considered the safer approach, as it minimizes the risk of immune incompatibility. Further research is required to better define the safety profile, indications, and long-term outcomes of allogeneic PRP applications.
-
Under current development trajectories, PRP has the potential to become a more cost-effective and widely accessible adjunct to existing treatment methods, particularly in cases that are traditionally difficult to manage. As protocols continue to be refined and standardized, PRP may see broader integration into surgical and interventional settings to support healing and tissue recovery.
Future applications could include expanded use alongside conventional therapies to enhance postoperative healing, support management of challenging conditions, and potentially improve tissue integration in select transplant or grafting procedures. Continued research will be necessary to define appropriate indications, optimize preparation methods, and establish long-term clinical outcomes.
